
Peter Zeihan: What Would a Conflict in Taiwan Look Like?
Let’s discuss what China’s potential invasion of Taiwan would look like. Should China attack, both Biden and Trump have been explicit that the US would…
Thought Leader: Peter Zeihan
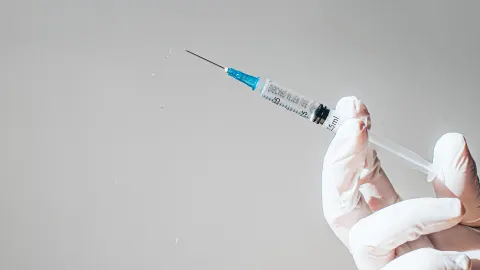
The US Food and Drug Administration has approved a vaccine against the respiratory syncytial virus, or RSV, for adults 60 and older. This vaccine, called Arexvy, is made by the drug company GSK. A recommendation for its use could come from the US Centers for Disease Control and Prevention as early as June, which means it could be available starting in the fall.
How important is a vaccine against RSV? How effective is the Arexvy vaccine? Could the vaccine be available this winter, and how can people access it? Do people still need to take other vaccines against the flu and Covid-19? And can this vaccine be used for young children who are also vulnerable to RSV?
To answer these questions, I spoke with CNN Medical Analyst Dr. Leana Wen. Wen is an emergency physician and professor of health policy and management at the George Washington University Milken Institute School of Public Health. She previously served as Baltimore’s health commissioner.
CNN: How important is it to have a vaccine against RSV?
Dr. Leana Wen: RSV is an extremely common virus, especially among young children. According to the CDC, virtually all children will have gotten it by the age of 2.
Most people who contract RSV will have mild cold-like symptoms. The problem is when RSV extends to the lower airway. The virus could cause bronchiolitis, an inflammation of the small airways of the lungs, and can extend to pneumonia, an infection of the lungs.
Around 58,000 to 80,000 children younger than 5 years old are hospitalized in the United States each year because of RSV, according to the CDC. For babies younger than 6 months of age, the rate of hospitalization is 1 to 2 out of every 100 kids infected with RSV. The risk for severe illness is highest among premature infants and children with underlying medical conditions, such as weakened immune systems.
Though RSV is most often discussed as a threat to children, it also poses a health risk to older adults. In the US, an estimated 159,000 adults 65 and older are hospitalized each year due to RSV, and around 10,000 to 13,000 older adults die from an infection. Those at highest risk for severe outcomes are people with underlying health conditions such as heart or lung disease or who are immunocompromised.
In short, there is a significant burden of illness caused by RSV. Having a vaccine that can reduce the severity of disease is a major step forward and can save many lives every year.
CNN: How effective is this particular vaccine, Arexvy?
Wen: It appears to be effective. Researchers published the findings of their phase 3 clinical trial recently in the New England Journal of Medicine. This was a randomized control trial involving nearly 25,000 adults 60 and older. Half received a placebo and the other received one dose of the vaccine. Vaccine efficacy was about 83% at preventing lower respiratory tract disease caused by RSV. Among patients with underlying health conditions like heart and lung disease, the vaccine was 94% effective at preventing lower respiratory tract disease. It was also 94% effective at preventing severe disease.
These are very promising results. It remains to be seen how long protection from the vaccine lasts and whether and how often people may need to receive additional doses. So far, the vaccine’s protection appears to last at least a year.
Researchers are studying this issue of durability as well as monitoring for severe side effects. In the phase 3 clinical trial, the most common side effects were pain at the injection site and fatigue, which resolved quickly. There were no differences in serious side effects between the placebo and vaccine groups, though, as with all medicines, there needs to be continued monitoring after the vaccine is given to a wider population.
CNN: Could the vaccine be available this winter season? If so, how can people get it?
Wen: Now that the FDA has approved the vaccine, the CDC will meet to discuss it. The CDC’s external advisers are set to meet in June, and the CDC could issue its recommendation to the CDC director shortly after. The director then makes the decision.
If all that happens, Arexvy could be available this fall. I think it would be available in the same places as the flu and Covid-19 vaccines — in pharmacies and in certain doctors’ offices. We will need to see which groups the CDC recommends receiving this vaccine — whether it’s everyone 60 and older or only those with certain medical conditions.
CNN: Do people still need to take other vaccines against the flu and Covid-19?
Wen: Yes. The RSV vaccine specifically targets RSV. It does not replace other vaccines that target other pathogens. People should still receive the annual flu vaccine this fall, and should follow CDC guidance on when to receive another Covid-19 booster.
CNN: Can this vaccine be used for young children who are also vulnerable to RSV?
Wen: This particular vaccine by GSK has only been approved for older adults. However, there is an RSV vaccine being developed by the drug company Pfizer that is being tested in pregnant women for the purpose of conveying protection to the infant. These results are already available — the FDA’s vaccine advisers are meeting on Thursday to review them. However, the FDA may not make a decision on when to make the vaccine available for pregnant people until this summer.
Pfizer has also submitted an application to the FDA for its RSV vaccine to be used in older adults, and a few other drug companies are expected to have phase 3 trials completed this year, too.
Peter Zeihan: What Would a Conflict in Taiwan Look Like?
Let’s discuss what China’s potential invasion of Taiwan would look like. Should China attack, both Biden and Trump have been explicit that the US would…
Thought Leader: Peter Zeihan
Dr. Sanjay Gupta: Rethinking Health Rules We Grew Up With
“Drink your milk,” we are told as children. CNN Chief Medical Correspondent Dr. Sanjay Gupta examines our practice of continuing to drinking cow’s milk as…
Thought Leader: Sanjay Gupta
Erika Ayers Badan: Turning What Fuels You Into Business
Kerri Rosenthal is an artist, a businesswoman, a mom, not someone giving up her ambition (is that what we’re supposed to be doing at work…
Thought Leader: Erika Ayers Badan

